 Long-term protection against Herpes Zoster by the adjuvanted Recombinant Zoster Vaccine: interim efficacy, immunogenicity, and safety results up to 10 years after initial vaccination. Shingles vaccine: Shingrix will be the new funded brand. The adjuvanted recombinant zoster vaccine confers long-term protection against herpes zoster: interim results of an extension study of the pivotal phase 3 clinical trials ZOE-50 and ZOE-70. Immune senescence and vaccines to prevent herpes zoster in older persons. Varicella-zoster vaccine for the prevention of herpes zoster. A critical appraisal of ‘Shingrix’, a novel herpes zoster subunit vaccine (HZ/Su or GSK1437173A) for varicella zoster virus. Systematic review of incidence and complications of herpes zoster: towards a global perspective. Efficacy of the herpes zoster subunit vaccine in adults 70 years of age or older. 
Efficacy of an adjuvanted herpes zoster subunit vaccine in older adults. 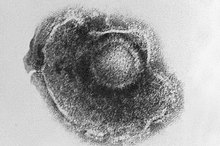
Lal H, Cunningham AL, Godeaux O, Chlibek R, Diez-Domingo J, Hwang S-J, et al.Prevention of herpes zoster: recommendations of the Advisory Committee on Immunization Practices (ACIP). Centers for Disease Control and Prevention.Adverse events involving GlaxoSmithKline products should be reported to GSK Medical Information on 0800 808 500. Marketed by GlaxoSmithKline NZ Ltd, Auckland. ©2023 GSK group of companies or its licensor. Additional product information and Consumer Medicine Information (CMI) is available at Trademarks are owned by or licensed to the GSK group of companies. If you have side effects, see your doctor, pharmacist or healthcare professional. Vaccination with SHINGRIX may not protect all vaccine recipients. Common side effects: Adults ≥50 years: pain, redness and swelling at the injection site, muscle pain, fatigue, headache, shivering, fever, and gastrointestinal symptoms. SHINGRIX has risks and benefits – ask your doctor if SHINGRIX is right for you. SHINGRIX should not be administered if you are hypersensitive to any component of this vaccine. A single 0.5 mL dose contains 50 micrograms of gE antigen, adjuvanted with AS01B (composed of the plant extract Quillaja saponaria saponin (QS-21) (50 mcg) and 3-O-desacyl-4’-monophosphoryl lipid A (MPL) from Salmonella minnesota (50 mcg) plus excipients). Costs will apply for people who are not 65 years old.  :max_bytes(150000):strip_icc()/shingles_symptoms_IL-5aec997c8e1b6e0039a1df31-f560011d41fa4a20907723462634c617.png)
SHINGRIX is a prescription medicine and is fully funded for individuals aged 65 years only. SHINGRIX (Recombinant Varicella Zoster Virus Glycoprotein E antigen 50 micrograms (AS01B adjuvanted vaccine)) is indicated for the prevention of herpes zoster (HZ) and post-herpetic neuralgia in adults 50 years of age or older and for adults 18 years of age or older who are at increased risk of herpes zoster.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
